Introduction
Pharmaceutical-grade purified water, often referred to as Purified Water (PW), is a critical component in pharmaceutical manufacturing and must meet stringent quality standards set by United States Pharmacopeia (USP) or European Pharmacopoeia (Ph. Eur.) or Indian Pharmacopeia. Purified water serves as a critical raw material for drug formulation, cleaning, and various laboratory applications. To ensure the safety and efficacy of pharmaceutical products, specific quality attributes of purified water must be controlled and monitored. In this blog, we will explore the critical quality attributes of purified water for pharmaceutical use, their significance, and how they are achieved.
What are Critical Quality Attributes (CQA) of PW?
Critical Quality Attributes refers to the specific parameters or characteristics that must be closely monitored and controlled to ensure water’s quality and suitability for pharmaceutical use. In order to ensure safety of the patients such key parameters are regulated by pharmacopeial bodies. Following are the critical quality attributes which
are important to be understood.
Purity: Purity is the foremost CQA for purified water in pharmaceuticals. It requires the absence of impurities such as organic and inorganic contaminants, microorganisms, endotoxins, and particulate matter. Any impurity can negatively impact the safety and effectiveness of drugs.
Chemical Composition: The chemical composition must be consistent and free from substances that could interact with pharmaceutical ingredients. Unwanted ions, residual chemicals, or heavy metals could lead to unwanted reactions in drug formulations.
Microbial Control: Maintaining low microbial counts is crucial. Microbial contaminants can lead to failure of drug sterility and impact safety of patients. Control is achieved through regular monitoring, sanitization, and validated water purification processes.
Endotoxin Levels: Endotoxins are cell wall components of certain bacteria that can cause severe pyrogenic reactions in humans. Purified water must have ultra-low endotoxin levels, usually achieved by specialized purification techniques.
Quality Parameters and Control limits
Pharmaceutical-grade purified water, must meet stringent quality standards set by regulatory agencies like the United States Pharmacopeia (USP) or European Pharmacopoeia (Ph. Eur.).
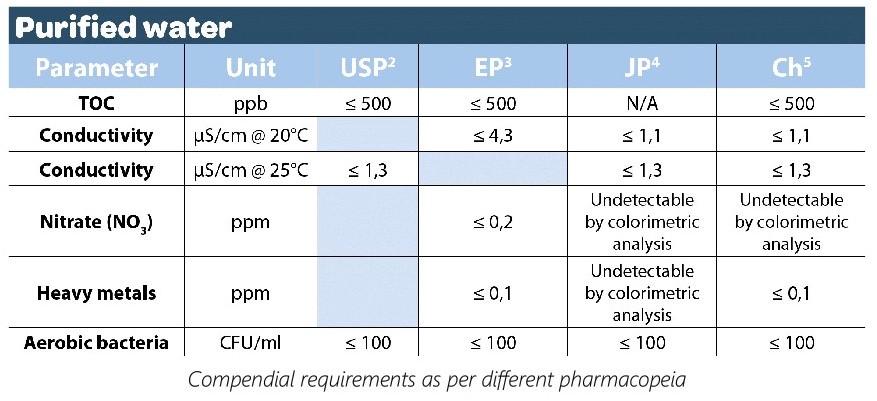
Total Organic Carbon (TOC): TOC measures the total amount of carbon in the water, which includes organic impurities. TOC levels in PW should be kept very low (typically less than 0.5 ppm) and are achieved through processes like activated carbon filtration and/or oxidation methods like UV or ozone treatment.
Chloride and Sulfate: Chloride and sulfate ions should be at very low levels to avoid interference with drug formulations. Ion-exchange resins and anion exchange membranes help reduce these ions to acceptable levels.
Conductivity: Low conductivity is essential to ensure the absence of ions in the water. Ion- exchange resins and reverse osmosis are used to reduce conductivity, typically aiming for less than 1.3 µS/cm.

Heavy Metals: Heavy metals like lead, cadmium, and mercury must be below specified limits (e.g., <0.1 ppm). Pretreatment with activated carbon and specific ion-exchange resins are generally used to remove heavy metals.
Microbial Contaminants: Purified water must be free of bacteria, fungi, and endotoxins. This is achieved through multiple stages of filtration (0.2 µm or smaller) and may include ultraviolet (UV) or ozone disinfection.
Particles: The water should be virtually free of particles, often requiring filtration through 0.2 µm or smaller pore size filters.
pH: PW typically has a pH range of 5.0 to 7.0, and it’s adjusted using purified acids or bases.
Resistivity: High resistivity (typically >18.2 MΩ·cm) is desired and is achieved through processes like reverse osmosis and deionization.

Endotoxins: Endotoxins (lipopolysaccharides from bacterial cell walls) must be reduced to very low levels, often through filtration and de- pyrogenation methods.
Why Critical Quality Attributes are important?
These critical quality attributes are vital for several reasons:
Patient Safety: Any impurities or contaminants in the water used in drug formulations can pose serious health risks to patients. Ensuring the water’s quality is crucial to prevent adverse reactions or contamination of the final drug product.
Drug Efficacy: The quality of water can affect the stability and efficacy of drug formulations. For instance, some drugs may degrade or become less effective in the presence of certain impurities or variations in water quality.
Regulatory Compliance: Regulatory authorities, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), have strict guidelines and regulations concerning the quality of pharmaceutical water. Failing to meet these standards can result in regulatory non-compliance, which may lead to product recalls, fines, or legal actions.
Consistency: Maintaining consistent water quality is vital for ensuring the reproducibility of pharmaceutical processes. Fluctuations in water quality can lead to variations in drug manufacturing, potentially affecting product quality and performance.
Process Integrity: Water is used in various pharmaceutical processes, including cleaning, formulation, and quality control testing. If the water used is not of the required quality, it can lead to inaccurate test results, cross-contamination, or inadequate cleaning, which can affect product safety and quality.
Cost Implications: Failing to meet the CQAs of purified water can result in increased production costs. Loss due to batch rejections, resolving the water quality issues or dealing with regulatory consequences can be costly and time-consuming.
How Critical Quality Attributes are achieved?
Achieving these critical quality attributes requires a comprehensive approach:
Water Purification Systems:
State-of-the-art water purification systems, such as reverse osmosis (RO) and distillation, are used to remove impurities.

Regular Testing: Routine testing of water samples is conducted for microbiological and chemical analysis. Advanced analytical instruments are employed for accurate measurements.
Validation and Documentation: Validation of purification processes, maintenance of records, and compliance with regulatory guidelines are essential components of achieving and maintaining CQAs.
Conclusion
In the pharmaceutical industry, the critical quality attributes of purified water are the foundation of drug safety and efficacy. Purity, chemical composition, microbial control, endotoxin levels, and conductivity must be rigorously monitored and maintained to meet regulatory standards and protect patients. The achievement of these attributes requires advanced purification technologies, stringent testing, and adherence to strict documentation and regulatory guidelines.
References
- United States Pharmacopeia (USP) Chapter <1231> Water for Pharmaceutical Purposes.
- European Pharmacopoeia (EP) Chapter 2.2.38 Water for Pharmaceutical Use.
- Japanese Pharmacopoeia (JP) General Information for Crude Drugs.
- U.S. Food and Drug Administration (FDA)
- European Medicines Agency (EMA)
- From study of the context to use of appropriate water treatment technologies https://www.a3p.org/en/from-study-of-the-context-to-use-of-appropriate- water-treatment-technologies/





















 WP Digitals
WP Digitals